News
Publications
The Rising Role of Radiotheranostics Supported by the Oncidium Foundation
By Richard Zimmermann and Rebecca Lo bue, SNMMI Value Initiative, July 2021.
Radiotheranostics Momentum
By Richard Zimmermann, SNMMI Value Initiative, January 2021.
Interest in Theranostics grows as capabilities advance
By Richard Zimmermann, HealthCare Business, June 2020.
Theranostics Insights
64Cu-SAR-Bombesin
Imaging
18F-FES
Imaging
67Cu-SARTATE
THERAPY
177Lu-FAP-2286
THERAPY
177Lu-NeoB
THERAPY
177Lu-PSMA-IT
THERAPY
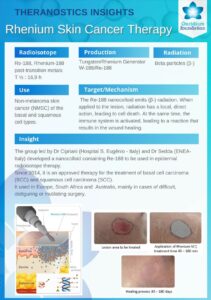
188Rhenium-SCT
THERAPY
225Ac-DOTATATE
THERAPY
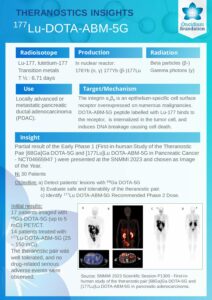
177Lu-DOTA-ABM-5G
THERAPY
177Lu-DOTA-ROSOPATAMAB
THERAPY
211At-BC8-B10
THERAPY
Videos
What Are Radiotheranostics for Cancer Care?
Doctor, will Radiotheranostics make me radioactive?
Tom, treated with PRRT for NETs
Fernando Acosta’s Testimonial with Lutetium-177 PSMA Radioligand Therapy
Clinical Trials
Participating in a Clinical Trial
- https://clinicaltrials.gov : US database of privately and publicly funded clinical studies conducted in the US and around the world.
- https://www.ancora.ai : Tool that can provide patients with innovative treatments in helping to find personalized trial options.
- https://www.theranostictrials.org: tool developed by Drs. Luke Nordquist and Oliver Sartor to provide Radiopharmaceutical clinical trials.
- https://euclinicaltrials.eu/search-for-clinical-trials/?lang=en : Clinical Trials Information System (CTIS). This website supports the undertaking and oversight of clinical trials in the European Union (EU) and European Economic Area (EEA).
- https://www.anzctr.org.au : ANZCTR is a register of clinical trials being undertaken in Australia and New Zealand.
- https://trialsearch.who.int/: the WHO International Clinical Trials Registry Platform imports data files from clinical registries from all over the world. It ensures that a complete view of research is accessible to all those involved in health care decision making.
Conducting a Clinical Trial
- Regulation:
- Clinical Trials Information System (CTIS) :
Under the Regulation, clinical trial sponsors can use the CTIS from 31 January 2022, but are not obliged to use it immediately, in line with a three-year transition period. National regulators in the EU Member States and EEA countries will use CTIS from 31 January 2022.
European Organisation for Research and Treatment of Cancer (EORTC):
Academic, independent, non-profit organisation under Belgian law, with no direct government subsidy. Clinical studies evaluating new drugs for potential registration, or testing therapeutic agents, are conducted in partnership with commercial organisations. This funding is subject to EORTC’s strict principles of independence.
The Clinical Research Initiative for Global Health (CRIGH):
CRIGH aims to optimize clinical research programs, develop global standards on clinical research, promote the take-up of innovative methodology and technologies, and encourage international cooperation to rapidly and efficiently respond to global health challenges
The European Clinical Research Infrastructure Network (ECRIN):
Non-profit intergovernmental organisation that supports the conduct of multinational clinical trials in Europe. As of 2013, ECRIN has the legal status of a European Research Infrastructure Consortium (ERIC). Works with European Correspondents across Europe, national networks of clinical trial units (CTUs), as well as European and international stakeholders involved in clinical research.
Links
Nuclear medicine
- The NME (Nuclear medicine Europe) has implemented a website for the general public called “What is Nuclear medicine ?”, that features movies, cartoons and special small booklets to be downloaded that explain basic concepts.
- Official bodies such as EANM and SNMMI also have materials explaining Nuclear medicine.
- You will find useful information as well on the IAEA website about Radiopharmaceuticals production and some interesting Publications regarding advances search.
Books
In order to explain this technology in an accessible way, a reference booklet containing information on Nuclear medicine was written by the author of this project. It was initially intended for workers in the Radiopharmaceutical industry but was then used for patients and finally distributed to potential investors. This book is presently available in French, English, and Spanish.