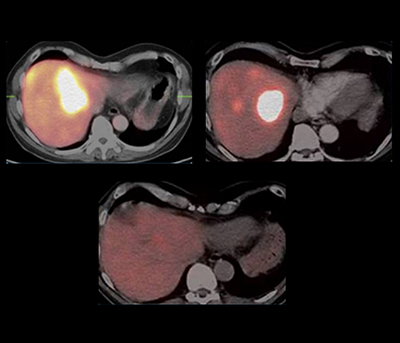
This series of three pictures illustrates in a very simple way the efficacy of such a treatment in neuroendocrine tumor (NET) patients (pancreatic cancer). The Lutetium-177 somatostatin analogue used in this patient has been obtained from the EMA under the brand name Lutathera.
(Courtesy of Prof. Dr. Richard P. Baum, Zentralklinik Bad Berka, Germany) Cure of liver metastases from neuroendocrine pancreatic cancer using intra-arterial peptide receptor Radionuclide therapy (PRRNT) shown here before treatment (on the left), one year after application of three cycles of Yttrium-90 somatostatin analogue (middle) and more than 5 years after the first image following the administration of an additional single systemic course of Lutetium-177 somatostatin analogue.
What it is?
Neuroendocrine tumors (NETs) are a diverse group of cancers that arise from neuroendocrine cells, which have features of both nerve cells and hormone-producing cells. NETs can occur throughout the body, but are most common in the gastrointestinal tract, pancreas, and lungs. NETs can grow slowly or more aggressively, depending on the type.
Because of this diversity, their classification is complex but crucial part of diagnosis and treatment planning. The most widely used system is the one developed by the World Health Organization (WHO), which classifies these tumors based on Grade, Differentiation, Site, and Hormone Production. Understanding the type of cancer is important because it helps guide the most effective treatment.
Mains Symptons
NETs can be tricky to detect because symptoms are often mild or mistaken for other illnesses. Common signs include:
- Belly pain or cramps
- Diarrhea or constipation
- Flushing (redness in the face or neck)
- Unexplained weight loss
- A fast heartbeat or feeling anxious
- High or low blood sugar levels (from hormone changes)
Some NETs don’t cause symptoms at all and are found by accident during tests for other illnesses.
Standard treatment
Treatment depends on where the tumor is, how fast it is growing, and if it has spread.
Common treatments include:
- Surgery to remove the tumor
- Somatostatin analogs medications that slow tumor growth and reduce hormone-related symptoms
- Chemotherapy used for faster-growing tumors, especially if the cancer has spread.
- Radiation therapy uses high-energy rays to kill cancer cells. Used if tumors are restricted to one area
- Targeted therapy used especially if the cancer has spread. Uses special drugs that attack specific parts of cancer cells, responsible for the cancer growth or survival; sparing most healthy cells.
- Liver-directed therapies if cancer spreads to the liver
Radiotheranostics
Radiotheranostics is a new type of treatment that combines therapy and diagnostics. It uses special radioactive substances that are injected in the patient, finds and destroys the cancer cells. One approved radiotheranostic option is 177Lu-DOTATATE. This treatment combines a radioactive particle (lutetium-177) with a targeting molecule that seeks out somatostatin receptors, which are often found on NET cells. Once bound, it delivers radiation directly to the cancer, helping shrink tumors and control symptoms while sparing most healthy tissue.
Your doctor can help you understand which treatment is right for you
To know more about:
Patient associations
International Neuroendocrine Cancer Alliance (INCA)
INCA represents the global voice of patients with neuroendocrine cancer and genetic neuroendocrine tumors (NETs), as well as their caregivers, and is compiling a list of NET support groups worldwide by country for outreach.
